In the spirit of both Christmas and recycling, I decided to build a Christmas tree by gluing together wine corks accumulated during this year. In addition, if you needed one, the intermediates of this process make rather cute models of polycyclic aromatic hydrocarbons.
Thursday, December 22, 2016
Tuesday, December 06, 2016
An electron wire in formylmethanofuran dehydrogenase
The first step of biological methane formation from carbon dioxide is the reduction of CO2 to form N-formylmethanofuran from methanofuran. This reaction is catalysed by formylmethanofuran dehydrogenase (EC 1.2.99.5). There are two types of this enzyme in methanogenic archaea, a tungsten iron—sulphur protein (Fwd) and a molybdenum iron—sulphur protein (Fmd).
Wagner et al. [1] determined the X-ray structures of a Fwd enzyme from the thermophilic methanogenic archaeon Methanothermobacter wolfeii in several crystal forms [2—4]. To any bioinorganic chemist this metalloprotein should look like a treasure trove: every FwdABCDFG heterohexamer has got a mononuclear tungsten centre, a dinuclear zinc centre, and quite a few iron—sulphur clusters. The enzyme exists as either a dimer or a tetramer of the FwdABCDFG heterohexamers. The authors suggest that the 24-meric complex (FwdABCDFG)4 is a physiologically active form. It contains 46 (yes, forty-six) [Fe4S4] clusters which form an electron wire between the redox-active tungsten centres. The function of this wire remains unclear though.


- Wagner, T., Ermler, U. and Shima, S. (2016) The methanogenic CO2 reducing-and-fixing enzyme is bifunctional and contains 46 [4Fe-4S] clusters. Science 354, 114—117.
- PDB:5T5I
- PDB:5T5M
- PDB:5T61
Sunday, July 31, 2016
A Guide to Psychoactive Plants
Humans were consuming, growing and trading (in this order) the psychoactive plants and derived substances since time immemorial. Most governments tried (and failed) to control and restrict them. Without these plants, not only pharmacology as we know it would not exist, but the whole human history would be completely different. Surely Guía de las plantas psicoactivas by Dr. Josep Lluís Berdonces i Serra [1], published by Ediciones Invisibles (I am not joking) is not the first and not the last book dealing with this topic. Why would we need another one? That’s exactly the question Jonathan Ott, the author of classic Pharmacotheon [2], asks (and answers) in the preface, which also mentions such classics as Plants of the Gods [3] and The Encyclopedia of Psychoactive Plants [4]. As for me, I just saw this beautifully illustrated book on display in the library and felt compelled to borrow it. It is written in a lively, easy-to-read Spanish. For such a relatively slim volume (333 pages including appendices and index), it’s surprisingly informative. I learned that...
- Purple morning glory, Ipomoea purpurea, is known in Spanish as don Diego de día;
- The popular decorative plant formerly known as Coleus blumei (now Plectranthus scutellarioides) is a mild hallucinogen;
- Saffron crocus, Crocus sativus, as well as the ice plant, Mesembryanthemum crystallinum, a succulent very common in Canary Islands, have an aphrodisiac effect.
It also contains a short chapter on psychoactive fungi and another one on pharmacology of principal active compounds, including (o joy!) their structural formulae. Perhaps inevitably, there are some omissions (which I hope will be addressed in the subsequent editions). For example, Guía dedicates enough space to coffee and kola, but where is tea? To fix that oversight, we’ve published our own short guide to psychoactive plants illustrated by Tamara Kulikova.
In Dr Ott’s view, this book “viene a expandir nuestros horizontes” (came to expand our horizons) — without the necessary consumption of its protagonists. With a cup of tea, maybe.

- Berdonces i Serra, J.L. (2015) Guía de las plantas psicoactivas: Historia, usos y aplicaciones. Ediciones Invisibles, Barcelona (ISBN 978-84-944195-4-6).
- Ott, J. (1993) Pharmacotheon: Entheogenic Drugs, Their Plant Sources and History. Natural Products Company.
- Schultes, R.E., Hofmann, A. and Rätsch, C. (2005) Plants of the Gods: Their Sacred, Healing, and Hallucinogenic Powers, 2nd Ed., Healing Arts Press, Rochester.
- Rätsch, C. (2005) The Encyclopedia of Psychoactive Plants: Ethnopharmacology and Its Applications, Park Street Press, Rochester.
Thursday, June 16, 2016
The end of unun*iums is announced
So that’s it, then. Four new elements, ununtrium, ununpentium, ununseptium and ununoctium [1] are to be officially named nihonium (symbol Nh), moscovium (Mc), tennessine (Ts), and oganesson (Og), respectively [2]. So that for a while the periodic table will be free of ungainly “unun” names. The provisional recommendation is out [3], comments by 8 November 2016. I am going to send mine to IUPAC, but I’d like to share them with my readers first.
Let’s start with naming conventions [4]:
In keeping with tradition, elements are named after:Unfortunately, it is not required for the names to be aesthetically pleasing.
- a mythological concept or character (including an astronomical object),
- a mineral, or similar substance,
- a place, or geographical region,
- a property of the element, or
- a scientist.
In absence of minerals (all four elements are artificial) and properties (apart from half-lives, which aren’t that long) to speak about, we are left with three options. Don’t you agree that the option (a) is the most interesting one? However, the discoverers have chosen easy and boring options (c) and (e). Well, I like nihonium, named after Nihon (ニホン), one of the Japanese names for Japan (literally, “the sun’s origin”). I can’t say the same about the rest.
Take moscovium:
It is proposed that the name moscovium and symbol Mc are given to element 115. Moscovium is recommended in recognition of the Moscow region and honoring the ancient Russian land that is home to the Joint Institute for Nuclear Research, where the discovery experiments were conducted using the Dubna Gas-Filled Recoil Separator in combination with the heavy-ion accelerator capabilities of the Flerov Laboratory of Nuclear Reactions, JINR.I see. Not content with dubnium (element 105), Russian scientists™ insist on honouring Dubna one more time. But Dubna is not Moscow. The town is at least as old as the Russian capital and is situated on the very edge of Moscow Oblast, or Podmoskovye (Подмосковье). Shouldn’t dubnium 2.0 be called podmoskovium then? And why mention Moscow at all? After all, there are four elements named after Ytterby. The variations like dubinium, dubonium, or poddubnium spring to mind. Needless to say, “Mc” will have to go.
Personally, I would prefer the element 115 to be named lemmium in honour of the late Motörhead frontman Lemmy Kilmister. Alas, IUPAC’s set of absurd rules (see above) restricts people after whom the new elements could be named to “scientists”. I’ll come to that in a minute.
Now let’s look at tennessine. The ending -ine, by analogy with English names of other halogens, appears to be natural. However it simply shows that the authors of this proposal (from Tennessee region, I guess) did not think about languages other than English, although they should have, for “the names for new chemical elements in English should allow proper translation into other major languages” [3]. For instance, in Latin the halogen names are fluorum, clorum, bromum, iodum and astatum, in Spanish they are flúor, cloro, bromo, yodo and astato, while in German they are simply Fluor, Chlor, Brom, Iod und Astat. So in these languages the element 117 must be named tennessum, teneso and Tenness, respectively.
As for the symbol, I was about to complain that Ts is is a bad choice for an element symbol as Ts is widely used for tosyl group, and why not to use Tn given that TN is also an abbreviation for Tennessee. There was a similar story with copernicium a few years ago (originally proposed symbol Cp was later changed to Cn). It’s not that the authors of the recommendation aren’t aware of potential confusion:
NB: We are aware of the fact that Ts is often used as abbreviation for the tosyl chemical group. However, this was not considered to be a valid objection, given the fact that we also use the symbols Ac and Pr for chemical elements, while chemists also use these as abbreviations for the acyl and the propyl groups. Very common items like AcOH and PrOH are usually not taken for the hydroxides of actinium and praseodymium and a possible confusion with the tosyl group seem extremely low. On the other hand, the abbreviation Tn, that might have been a natural suggestion, is impossible given the earlier (1923) CIAAW-IUPAC acceptance of that symbol for thoron (220Rn), and its regular usage since then, see e.g. Journal of Environmental Radioactivity.Still, I don’t find this convincing. If we ever get enough of ununseptium, we’ll find that its chemistry is nothing like that of actinium or praseodymium. Think of tosyl chloride, abbreviated TsCl. Now think of its tennessine analogue, abbreviated TsTs. That’s just silly. It’s a shame we can’t use “Tn” as TN is an abbreviation for the state of Tennessee. What about Tq then, after Tanasqui, the first recorded version of this toponym?
Finally, oganesson. Does it have to end with -on? Yes, most noble gases do (and, in contrast to meaningless -ine, the ending -on is present in other languages). Except helium, that is. Helium was named after Helios, the Greek god of the Sun, and is the only noble gas following the naming principle (a). Several “on” names are of Greek origin, namely νέον “new”, ἀργόν “inactive”, κρυπτόν “hidden” and ξένον “foreign”, whereas radon is a contraction of “radium emanation”. But oganesson... Please! It’s two syllables too many and feels out of place.
And another thing. I don’t know about you, but naming anything after a living person makes me uneasy. Seeing his surname mutilated this way should make Yuri Oganessian uneasy too. Come on, did we ran out of deserving dead scientists whose names, incidentally, can be used for the heaviest known element? For example, we can honour John Dalton, an English polymath best known for development of atomic theory; among other things, he invented his own symbols for chemical elements [5]. Or J. J. Thomson, discoverer of the electron. Or Francis W. Aston, discoverer of many naturally occurring isotopes. Or Arthur Compton, known for Compton scattering. Or C. T. R. Wilson, inventor of the cloud chamber. Or Fritz London, after whom the London dispersion forces are named. Or Ernest Walton, the first person to artificially split the atom.
To summarise: political considerations, inflated egos, and lack of imagination all may be responsible for some of the dismal proposals above. It does not mean we have to swallow them without fight. If you have better suggestions — and I’m sure you do — I urge you to write to IUPAC before 8 November.
- IUPAC announces the verification of the discoveries of four new chemical elements: The 7th period of the periodic table of elements is complete. IUPAC Press Release, 30 December 2015.
- IUPAC is naming the four new elements nihonium, moscovium, tennessine, and oganesson. IUPAC Press Release, 8 June 2016.
- Öhrström, L. and Reedijk, J. (2016) Names and symbols of the elements with atomic numbers 113, 115, 117 and 118. IUPAC Provisional Recommendation.
- Koppenol, W.H., Corish, J., García-Martínez, J., Meija, J. and Reedijk, J. (2016) How to name new chemical elements (IUPAC Recommendations 2016). Pure and Applied Chemistry 88, 401—405.
- Dalton, J. (1808) A New System of Chemical Philosophy, vol. I.
Sunday, April 03, 2016
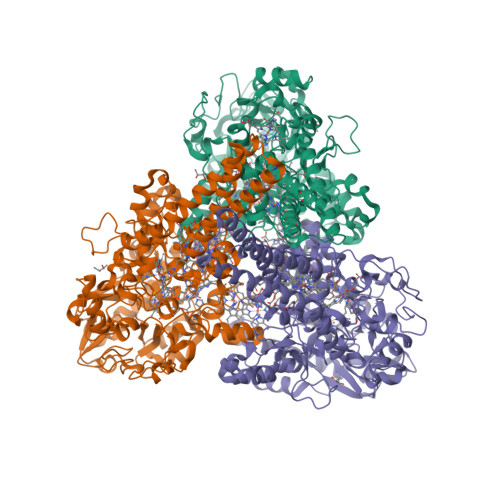
Octahaem sulphite reductase MccA
The epsilonproteobacterium Wolinella succinogenes is able to grow by sulphite respiration with formate as electron donor [1], thanks to the octahaem cytochrome c MccA that catalyses the six-electron reduction of sulphite to sulphide:
The crystal structure of MccA has been determined at 2.2 Å resolution [2, 3]. The enzyme exists as a homotrimer showing a novel fold and haem arrangement. The heterobimetallic active centre contains a Cu(I) ion and a haem c with a Fe—Cu distance of 4.4 Å [4].
|
- Kern, M., Klotz, M.G. and Simon, J. (2011) The Wolinella succinogenes mcc gene cluster encodes an unconventional respiratory sulphite reduction system. Molecular Microbiology 82, 1515—1530.
- PDB:4RKM
- PDB:4RKN
- Hermann, B., Kern, M., La Pietra, L., Simon, J., Einsle, O. (2015) The octahaem MccA is a haem c-copper sulfite reductase. Nature 520, 706—709.
Saturday, February 27, 2016
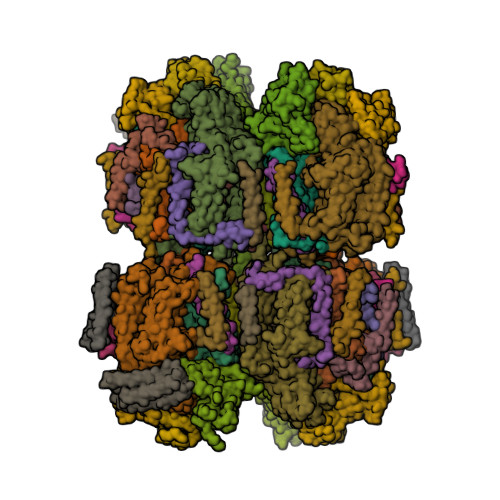
First eukaryotic photosystem II solved at 2.76 Å
The water-splitting reaction of photosynthesis is catalysed by photosystem II (PSII), a large protein complex located in thylakoid membranes of organisms ranging from cyanobacteria to higher plants [1]. During the last 15 years, a number of crystal structures of PSII from cyanobacteria have been solved. However, no structures of PSII from eukaryots have been reported until now, partly due to the instability of eukaryotic PSII upon isolation. Ago et al. [2] solved the structure of PSII from a red alga Cyanidium caldarium at 2.76 Å resolution [3]. This PSII contains four extrinsic proteins, including the three subunits found in cyanobacterial PSII and the fourth subunit PsbQ' homologous to the PsbQ protein of green algae and higher plants. Furthermore, two novel trans-membrane helices were found in the algal PSII which are not present in cyanobacterial PSII.
- Shen, J.-R. (2015) The structure of photosystem II and the mechanism of water oxidation in photosynthesis. Annual Review of Plant Biology 66, 23—48.
- Ago, H., Adachi, H., Umena, Y., Tashiro, T., Kawakami, K., Kamiya, N., Tian, L., Han, G., Kuang, T., Liu, Z., Wang, F., Zou, H., Enami, I., Miyano, M. and Shen, J.-R. (2016) Novel features of eukaryotic photosystem II revealed by its crystal structure analysis from a red alga. J. Biol. Chem. 291, 5676—5687.
- PDB:4YUU
Saturday, January 30, 2016
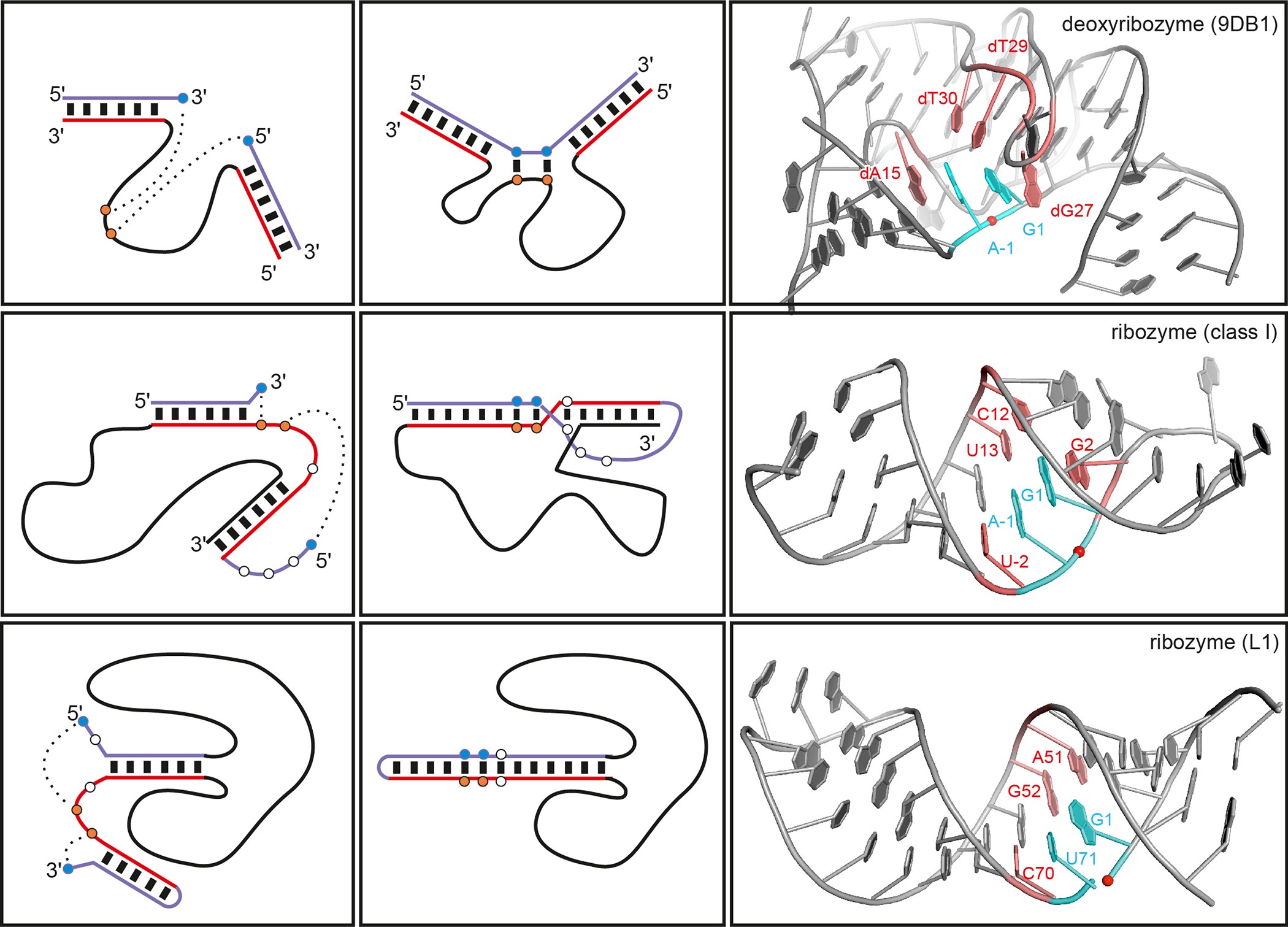
Crystal structure of the DNAzyme 9DB1
The first ever crystal structure of a deoxyribozyme has been solved at 2.8 Å resolution [1—3]. The work by researchers from Max Planck Institute for Biophysical Chemistry (Göttingen, Germany) also sheds light on a difference in catalytic mechanism of ribozymes and deoxyribozymes [4]:
Ribozymes use RNA’s 2´-hydroxyl groups, which are absent in DNA, for structural interactions or directly for catalysis. The new structure shows why the lack of these groups doesn’t diminish the catalytic activity of DNAzymes. The missing hydroxyls make DNA’s sugar-phosphate backbone more flexible, allowing acrobatic conformations that compensate for the absent hydroxyls in DNAzymes.
- Ponce-Salvatierra, A., Wawrzyniak-Turek, K., Steuerwald, U., Höbartner, C. and Pena, V. (2016) Crystal structure of a DNA catalyst. Nature 529, 231—234.
- PDB:5CKK
- PDB:5CKI
- Borman, S. (2016) After two decades of trying, scientists report first crystal structure of a DNAzyme. Chemical & Engineering News 94, issue 2, p. 3.








