Following the call to the community from Antony Williams, I indulged in some chemical drawing. I did redraw structure 1 from this paper from scratch to get (a). This is very much like structure of digitonin in ChEBI (b), except for methyl group at C-20 which goes up in (a).
 (a) | 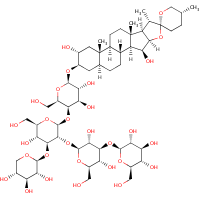 (b) |
Muhr et al. wrote:
With our investigations, it was possible for the first time to confirm beyond all doubt the structure suggested by Tschesche and Wulff for digitonin by means of modern NMR techniques, and to assign all proton and carbon resonances.
Now I was not able to get to the full text of Tschesche and Wulff, but at least their abstract contains the German name “3[β-D-Glucopyranosyl(I)(1→3Galakt.II)-β-D-galaktopyranosyl(II)(1→2Gluc.III)-β-D-xylopyranosyl(1→3Gluc.III)-β-D-glucopyranosyl(III) (1→4Galakt.IV)-β-D-galaktopyranosyl(IV)(1→3-Digitog.)]5α,20βF,25α Spirostantriol(2α,3β,15β)”, which kind of confirms 20β configuration. (The default configuration of spirostan is 20α.)
I guess this still does not answer what the “correct” structure of digitonin is. All we can say that Muhr et al. reported the structure (a).







No comments:
Post a Comment